# Intro to Optogenetics
Matthew Q. Clark
## Introduction to Optogenetics in Drosophila Larvae:
In this lab, we will explore the use of optogenetics, a technique that
allows precise control of neuronal activity using light. By employing
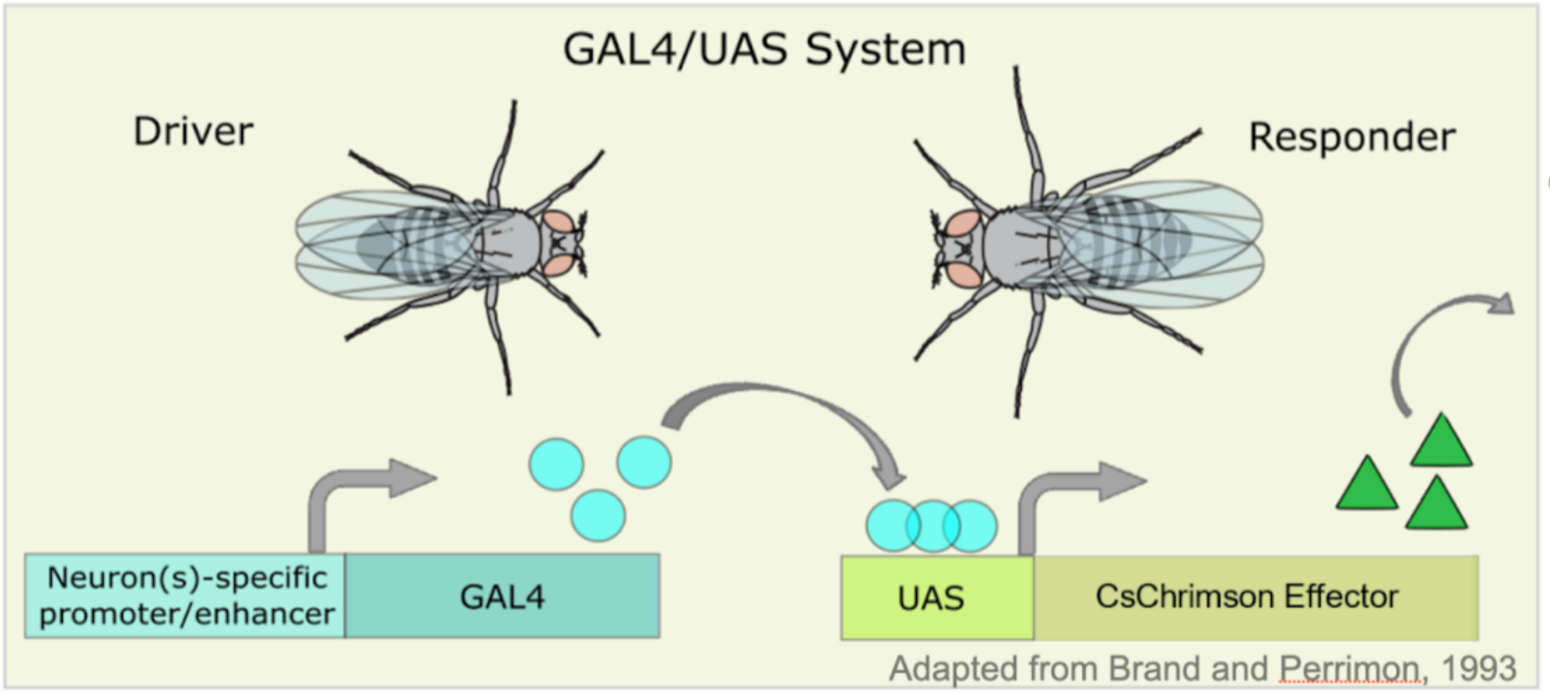
the GAL4/UAS system
(Figure 1 (a)), we can
target specific neurons in *Drosophila melanogaster* larvae to express
light-sensitive proteins like
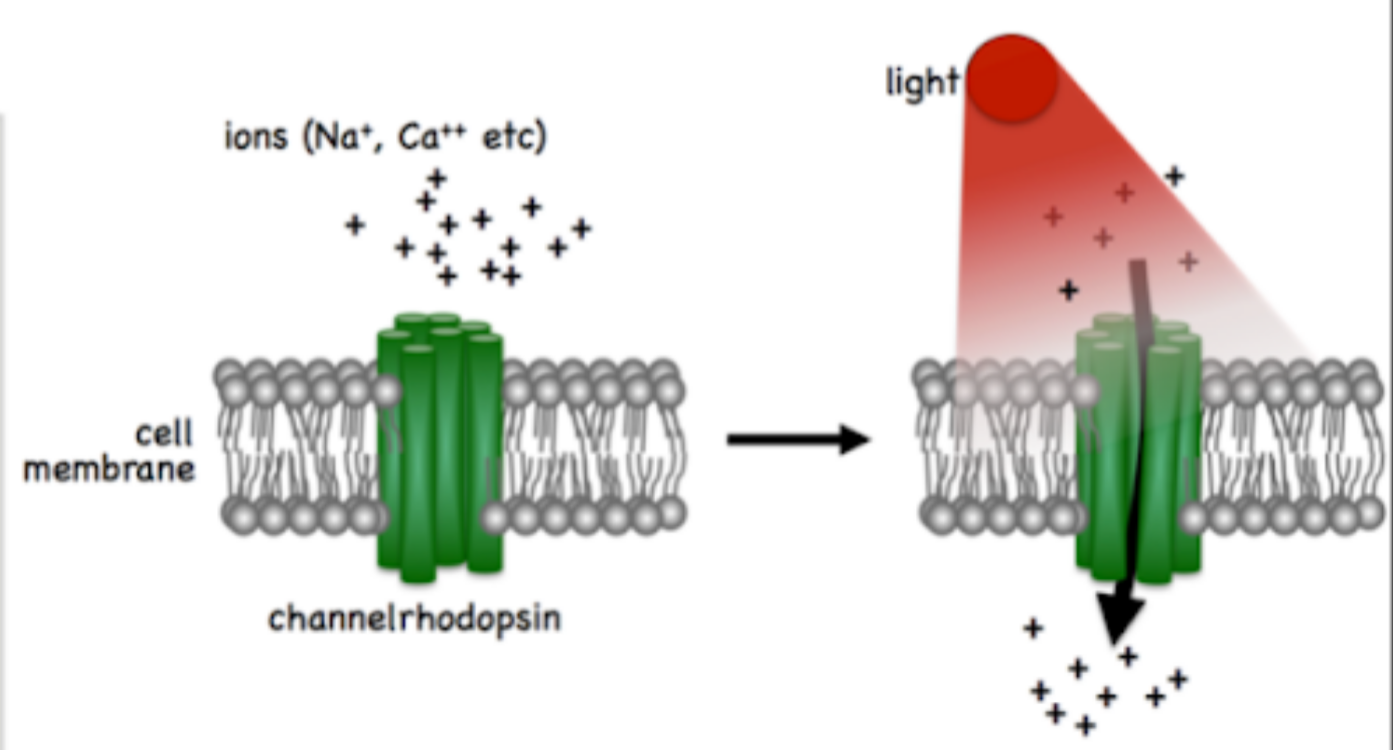
channelrhodopsins(Figure 1 (b)).
When exposed to specific wavelengths of light, these channelrhodopsins
open ion channels, leading to neuronal activation or inhibition. Through
this approach, we can observe and compare the resulting behavioral
changes in experimental larvae, which have been genetically modified,
against control larvae that have not. This experiment offers a hands-on
opportunity to understand the functional role of specific neurons in
controlling behavior.
(a)
(b)
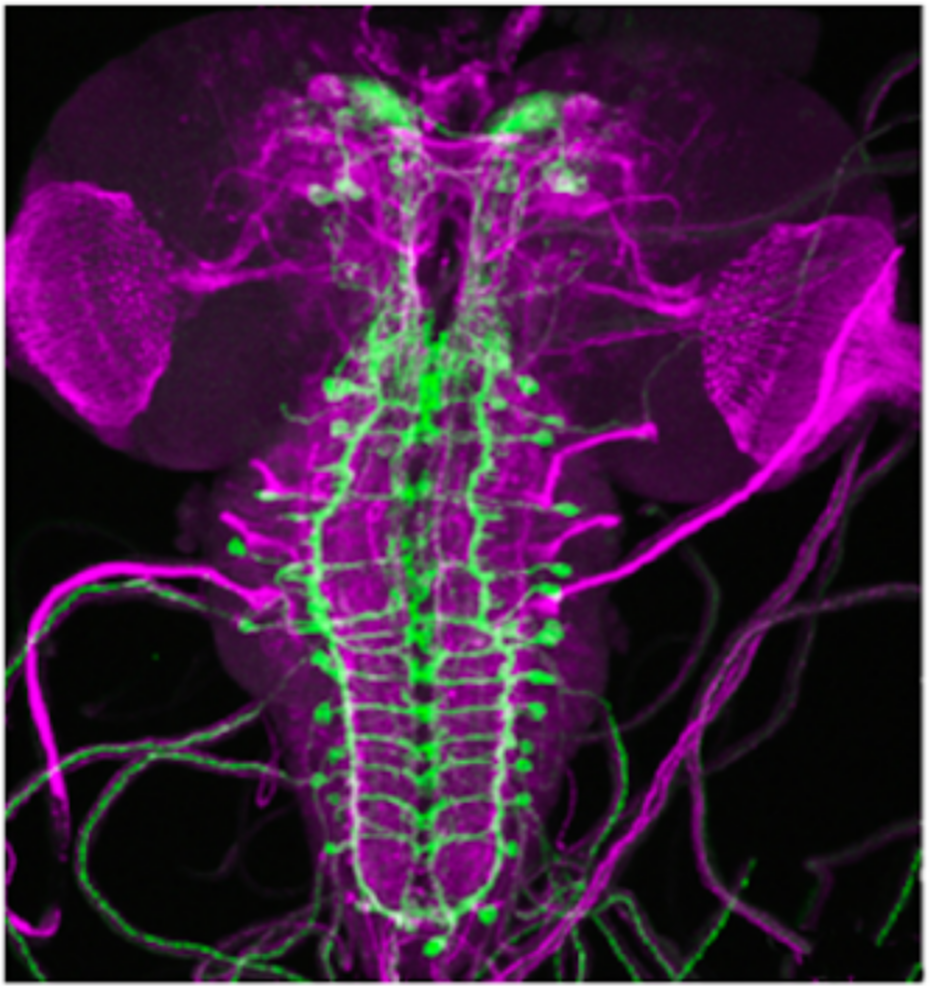
(c)
Figure 1: Optogenetics
### Materials
- Drosophila crosses: Control and experimental flies
- Leica EZ4 stereo microscope
- water, paintbrush
- Behavior arenas
### Protocol
1. Connect and turn on the LED light source. Set up the Leica EZ4
stereo microscope with all 3 LED illumination then turn it off via
the power switch in the back.
2. Warm to room temperature grape juice agar plates.
3. Collect a transgenic larval Drosophila expressing Channelrhodopsin
and transfer an individual to the agar assay plate.
4. Allow larva to acclimate for ~1-2 minutes and then observe it
crawling with the light off.
5. To stimulate neurons, turn on all 3 LED illumination for 4 seconds,
and record the number of peristalic waves. Let the larva rest for 10
seconds and repeat 2 more times for a total of 3 trials. Repeat this
for 4 additional larvae so the number of samples is 5 total.
6. For quantification, score for unique evoked phenotypes such as
pausing, turning and reverse crawling behaviors during periods of
LED stimulation versus no stimulation.
7. Compare rates of behaviors between stimulated and non-stimulated
periods to evaluate effects of optogenetic stimulation on larval
crawling. Compare rates of the control vs. the experimental. How are
they similar? How are they different? Is one better to compare
vs. the other?
8. Use excel/sheets to record your data and quantify and graph your
results (hint: if you’re not sure try uploading it to
[estimationstats.com](https://www.estimationstats.com/) to graph
your results!
 (a)
(a)
 (b)
(b)
 (c)
(c)
 (a)
(a)
 (b)
(b)
 (c)
(c)